Collaborators:
Ellie Lupton, Paige Carmen, Wisam Salah
Introduction and Problem:
Over the last 250 years, ocean acidification has increased at a faster rate than in any point in history. The issue: carbon dioxide. Due to the vast amounts of carbon dioxide that humans are pumping into the atmosphere, the pH of the ocean has risen from 8.1 to 8.2. Though this amount may not seem to be that impactful, the ocean must remain at a specific balance of chemicals and pH level in order to support life. "The ocean absorbs about a quarter of the CO2 we release into the atmosphere every year, so as atmospheric CO2 levels increase, so do the levels in the ocean" (PMEL, NOAA). The change in pH in the past years has caused a huge decrease of the amount of calcium carbonate, which is utilized by many marine organisms to build shells. Most notable of these organisms are the astounding coral that attract scuba diving and snorkeling tourists from around the world. The coral and other similar organisms are not able to grow or even rebuild themselves after being injured, and are slowly fading. Unless humans alter their actions and reduce their carbon footprint, biodiversity in the oceans will continue to plummet, until only the hardiest of organisms are able to survive their acidic depths. This lab will aid in addressing the question, "How does carbon dioxide affect the pH of ocean water, and in turn, how does this change in pH affect the delicate balance of life in the ocean?" By answering this through specific procedures and tests, one will be able to come to conclusions about the overall effect of ocean acidification on the biodiversity of the ocean as a whole.
Hypothesis:
If carbon dioxide is pumped into samples of distilled water and ocean water, then the pH of the salty ocean water will decrease sharply and cause the water to become much more acidic while the distilled water will remain relatively the same.
Parts of an Experiment:
Materials:
Methods and Procedure:
1. Use a graduated cylinder to measure 10 mL of ocean water. Pour it into test tube #1. Add about 10 drops of universal indicator. Stir and record pH in the data table.
2. Use a graduated cylinder to measure 10 mL of distilled water. Pour it into test tube #2. Add about 10 drops of universal indicator. Stir and record pH in the data table.
3. Blow up a large balloon and secure the opening with a twist tie (Hint: twist the balloon neck before securing opening).
4. Take a straw and insert into the balloon neck.
5. Place the tip of the straw in the ocean water sample. Undo the twist tie and slowly release pressure on the balloon's neck.
6. Use a stopwatch to start timing as soon as the pressure is released and the solution begins bubbling. Stop timing as soon as the color changes. Record your data in the table.
7. Repeat the procedure using the sample of distilled water and record in the data table.
8. Add crushed calcium carbonate to the ocean water and distilled water test tubes. Record your results.
9. Thoroughly clean your work station including all cylinders and test tubes.
Data and Data Analysis:
Ellie Lupton, Paige Carmen, Wisam Salah
Introduction and Problem:
Over the last 250 years, ocean acidification has increased at a faster rate than in any point in history. The issue: carbon dioxide. Due to the vast amounts of carbon dioxide that humans are pumping into the atmosphere, the pH of the ocean has risen from 8.1 to 8.2. Though this amount may not seem to be that impactful, the ocean must remain at a specific balance of chemicals and pH level in order to support life. "The ocean absorbs about a quarter of the CO2 we release into the atmosphere every year, so as atmospheric CO2 levels increase, so do the levels in the ocean" (PMEL, NOAA). The change in pH in the past years has caused a huge decrease of the amount of calcium carbonate, which is utilized by many marine organisms to build shells. Most notable of these organisms are the astounding coral that attract scuba diving and snorkeling tourists from around the world. The coral and other similar organisms are not able to grow or even rebuild themselves after being injured, and are slowly fading. Unless humans alter their actions and reduce their carbon footprint, biodiversity in the oceans will continue to plummet, until only the hardiest of organisms are able to survive their acidic depths. This lab will aid in addressing the question, "How does carbon dioxide affect the pH of ocean water, and in turn, how does this change in pH affect the delicate balance of life in the ocean?" By answering this through specific procedures and tests, one will be able to come to conclusions about the overall effect of ocean acidification on the biodiversity of the ocean as a whole.
Hypothesis:
If carbon dioxide is pumped into samples of distilled water and ocean water, then the pH of the salty ocean water will decrease sharply and cause the water to become much more acidic while the distilled water will remain relatively the same.
Parts of an Experiment:
- Independent Variable: Type of water used.
- Dependent Variable: Color of the universal indicator.
- Controlled Variables: Types of test tubes and graduated cylinders used, balloons, straw, room temperature, humidity, amount of carbon dioxide, amount of water, and amount of universal indicator (10 drops).
- Control Group: 10 mL of distilled water with the universal indicator.
- Experimental Group: 10 mL of ocean water with the universal indicator.
Materials:
- Two test tubes
- Graduated Cylinder
- Pipette
- Universal Indicator
- Ocean water (this is real ocean water, thanks to Mrs. Bostic)
- Distilled water
- Two balloons
- Straw
Methods and Procedure:
1. Use a graduated cylinder to measure 10 mL of ocean water. Pour it into test tube #1. Add about 10 drops of universal indicator. Stir and record pH in the data table.
2. Use a graduated cylinder to measure 10 mL of distilled water. Pour it into test tube #2. Add about 10 drops of universal indicator. Stir and record pH in the data table.
3. Blow up a large balloon and secure the opening with a twist tie (Hint: twist the balloon neck before securing opening).
4. Take a straw and insert into the balloon neck.
5. Place the tip of the straw in the ocean water sample. Undo the twist tie and slowly release pressure on the balloon's neck.
6. Use a stopwatch to start timing as soon as the pressure is released and the solution begins bubbling. Stop timing as soon as the color changes. Record your data in the table.
7. Repeat the procedure using the sample of distilled water and record in the data table.
8. Add crushed calcium carbonate to the ocean water and distilled water test tubes. Record your results.
9. Thoroughly clean your work station including all cylinders and test tubes.
Data and Data Analysis:
|
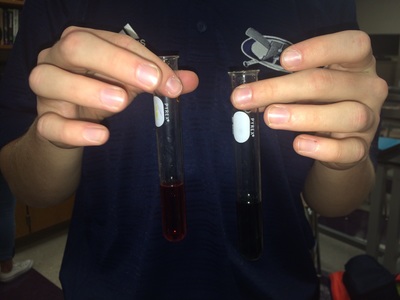
Photographs:
Images courtesy of: Paige Carmen |
Data:
Solution Measured pH Measured pH after adding CO2 Time for pH to change Observation with calcium carbonate
Ocean Water 9 7 6.3 seconds stayed relatively same pH now 8
Distilled Water 7 2 2.5 seconds became more neutral pH now 6
________________________________________________________________________________________________________________________________________________________________________
Analysis:
Two contrasting varieties of water, ocean water and distilled water, were placed into separate test tubes to have their pH levels measured after adding carbon dioxide and calcium carbonate. After organizing the data using a table, some outright questions arose as it became evident that the ocean water took about twice the time to change color than that of distilled water but the ocean water had a less dramatic change than distilled water. Rather, our assumptions of how each water sample would react to the addition of carbon dioxide were quite the opposite. Although the amount of time it took for the pH to change in the ocean water was high, the measured pH after adding CO2 was hardly nothing at all. This is most likely because the ocean already has certain amounts of CO2 within its water molecules, thus the alterations in pH levels were slim. On the other hand, the distilled water began the test with a measured pH of 7, neutral. As the balloon added carbon dioxide to the test tube, an immediate color change occurred. Another qualitative observation was gathered during the calcium carbonate test. The calcium carbonate tended to stay at the bottom of the solutions, causing the bottom to become a darker (more basic) color than the upper portion of the test tube with solution in it. As seen in photo 9, after the calcium carbonate was added to the distilled water, a few bubbles remained at the top of the solution. This was the predicted outcome since calcium carbonate is a pH buffer however, because the ocean water did not return to its original pH many organisms in that ecosystem would still be heavily effected. One thing that would improve this specific experiment's overall conclusion would be to discover the verified pH of the universal solvent. When adding too much of the solvent the data becomes distorted so knowing the repercussions of this particular solvent on the pH of both water samples would all in all enhance the analysis.
Conclusion:
Final results of this experiment denied the hypothesis that if carbon dioxide is pumped into samples of distilled water and ocean water, then the pH of the salty ocean water will decrease sharply and cause the water to become much more acidic while the distilled water will remain relatively the same. One may conclude that oceans absorbing 25-30% of human-induced carbon dioxide is beneficial to the Earth because it decreases the amount that is released into the atmosphere. This is not necessarily the case, according to some scientists. "Ocean acidity, as measured by pH, has increased by 30% since the industrial revolution and scientists predict pH will continue to change as increasing amounts of carbon dioxide are absorbed by oceans" (Climate and the Carbon Cycle). Because the chemistry of the oceans is important to life, subtle changes in that chemistry may have significant effects on the health of individual species and on entire ecosystems. In fact, ocean acidification can be detrimental to certain species by hindering shell growth of marine organisms such as corals, shellfish, and crabs. While the most common pH of ocean water is around 8.1, the pH of the ocean water that we measured initially was 9. To have it measure more basic than normal, many things could have accounted for this. The most likely reason would have been that the test tubes were not washed well beforehand. There is also the possibility that some variable in the testing environment was significantly different than what it should be in an oceanic environment. The distilled water, in comparison to the ocean water, had a greater change in pH overall. The pH of the distilled water dropped from an initial 7 to a 2 after adding CO2, while the pH of the ocean water dropped from a 9 to an 8. This occurrence is most likely a result of the two test tubes of water being "contaminated" or invalid at the beginning of the experiment. They both, however, were supposed to level out at the same pH, which they did, as water can absorb the same amount of carbon dioxide, regardless of whether the water contains salt or not. Distilled water had a much more significant change in pH due to the fact that it has no initial CO2 within its water molecules while the ocean water sample already has CO2 within itself.
On the other hand, when calcium carbonate was added to the water samples, both samples became more basic. The pH of the distillled water rose from 2 to about 6 while the pH of the ocean water rose from 7 to 8. The ocean water reacted more to the calcium carbonate as a result of carbonate being the buffering system of the ocean. "An integral part of the carbonate system is the pH (acid-base) buffer of seawater" (cms.udel.edu). As the excess of carbon dioxide was added to the water, more hydrogen ions were released, which cause the calcium carbonate to become bicarbonate, which then helps the ocean water to become more basic, (to be exact a pH of 8.1), the level at which it should normally be.
Now, the argument of whether or not this experiment is a valid model of ocean acidification solely depends on what the goal of the experiment is. If one is trying to catch a glimpse of the general effects of carbon dioxide on the pH of the ocean, than this experiment works quite well. On the contrary, if one is trying to see the actuality of how carbon dioxide affects the ocean and its pH, this experiment does not quite fit the bill. For one, a person simply blowing into a balloon and the CO2 escaping through a straw into the ocean water will add quite a different percentage/amount of carbon dioxide into the water than would happen in the environment. The sole fact that the beginning pH of both waters was much lower than that of real world waters simply shows just how high the percent error can be in this particular experiment. Also, there was most likely saliva and rubber residue in the air that came out of the balloon which could have also effected the ph.
All inclusive, ocean acidification is dramatically increasing because of human related increases in carbon dioxide emissions, which makes it an important topic in our AP Environmental Science class because the emitted CO2 is mainly originated by humans and is having major effects on our Earth's overall well being. For instance, a Smithsonian Ocean Portal article and a Climate Interpreter article examine and scrutinize the impairment of ocean acidification on aquatic ecosystems and species, "so much carbon dioxide is dissolving into the ocean so quickly that this natural buffering hasn’t been able to keep up, resulting in relatively rapidly dropping pH in surface waters" (Smithsonian Ocean Portal) and "a more acidic environment has a dramatic effect on some calcifying species, including oysters, clams, sea urchins, shallow water corals, deep sea corals, and calcareous plankton" (PMEL, NOAA). Each article continues on to explain the overall impact that ocean acidification has on overall marine life and each helps in embellishing this realization. In the grand scheme of things, this experimental lab assists in showing that carbon dioxide does indeed have a discrete effect on the pH of water however, it fails to exhibit the attested span of the problem.
Citation(s):
"Lab 7: Ocean Acidification: Too Much of a Good Thing?" Climate and the Carbon Cycle. N.p., n.d. Web. 17 Apr. 2015. <http://serc.carleton.edu/eslabs/carbon/lab7.html>.
"Ocean Acidification." Smithsonian Ocean Portal. N.p., n.d. Web. 17 Apr. 2015. <http://ocean.si.edu/ocean-acidification>.
"Ocean Acidification." Ocean Acidification. N.p., n.d. Web. 17 Apr. 2015. <http://www.pmel.noaa.gov/co2/story/Ocean%2BAcidification>.
"Ocean Acidification." Ocean Acidification. N.p., n.d. Web. 17 Apr. 2015. <http://co2.cms.udel.edu/Ocean_Acidification.htm>.
By: Noah Nelson
Solution Measured pH Measured pH after adding CO2 Time for pH to change Observation with calcium carbonate
Ocean Water 9 7 6.3 seconds stayed relatively same pH now 8
Distilled Water 7 2 2.5 seconds became more neutral pH now 6
________________________________________________________________________________________________________________________________________________________________________
Analysis:
Two contrasting varieties of water, ocean water and distilled water, were placed into separate test tubes to have their pH levels measured after adding carbon dioxide and calcium carbonate. After organizing the data using a table, some outright questions arose as it became evident that the ocean water took about twice the time to change color than that of distilled water but the ocean water had a less dramatic change than distilled water. Rather, our assumptions of how each water sample would react to the addition of carbon dioxide were quite the opposite. Although the amount of time it took for the pH to change in the ocean water was high, the measured pH after adding CO2 was hardly nothing at all. This is most likely because the ocean already has certain amounts of CO2 within its water molecules, thus the alterations in pH levels were slim. On the other hand, the distilled water began the test with a measured pH of 7, neutral. As the balloon added carbon dioxide to the test tube, an immediate color change occurred. Another qualitative observation was gathered during the calcium carbonate test. The calcium carbonate tended to stay at the bottom of the solutions, causing the bottom to become a darker (more basic) color than the upper portion of the test tube with solution in it. As seen in photo 9, after the calcium carbonate was added to the distilled water, a few bubbles remained at the top of the solution. This was the predicted outcome since calcium carbonate is a pH buffer however, because the ocean water did not return to its original pH many organisms in that ecosystem would still be heavily effected. One thing that would improve this specific experiment's overall conclusion would be to discover the verified pH of the universal solvent. When adding too much of the solvent the data becomes distorted so knowing the repercussions of this particular solvent on the pH of both water samples would all in all enhance the analysis.
Conclusion:
Final results of this experiment denied the hypothesis that if carbon dioxide is pumped into samples of distilled water and ocean water, then the pH of the salty ocean water will decrease sharply and cause the water to become much more acidic while the distilled water will remain relatively the same. One may conclude that oceans absorbing 25-30% of human-induced carbon dioxide is beneficial to the Earth because it decreases the amount that is released into the atmosphere. This is not necessarily the case, according to some scientists. "Ocean acidity, as measured by pH, has increased by 30% since the industrial revolution and scientists predict pH will continue to change as increasing amounts of carbon dioxide are absorbed by oceans" (Climate and the Carbon Cycle). Because the chemistry of the oceans is important to life, subtle changes in that chemistry may have significant effects on the health of individual species and on entire ecosystems. In fact, ocean acidification can be detrimental to certain species by hindering shell growth of marine organisms such as corals, shellfish, and crabs. While the most common pH of ocean water is around 8.1, the pH of the ocean water that we measured initially was 9. To have it measure more basic than normal, many things could have accounted for this. The most likely reason would have been that the test tubes were not washed well beforehand. There is also the possibility that some variable in the testing environment was significantly different than what it should be in an oceanic environment. The distilled water, in comparison to the ocean water, had a greater change in pH overall. The pH of the distilled water dropped from an initial 7 to a 2 after adding CO2, while the pH of the ocean water dropped from a 9 to an 8. This occurrence is most likely a result of the two test tubes of water being "contaminated" or invalid at the beginning of the experiment. They both, however, were supposed to level out at the same pH, which they did, as water can absorb the same amount of carbon dioxide, regardless of whether the water contains salt or not. Distilled water had a much more significant change in pH due to the fact that it has no initial CO2 within its water molecules while the ocean water sample already has CO2 within itself.
On the other hand, when calcium carbonate was added to the water samples, both samples became more basic. The pH of the distillled water rose from 2 to about 6 while the pH of the ocean water rose from 7 to 8. The ocean water reacted more to the calcium carbonate as a result of carbonate being the buffering system of the ocean. "An integral part of the carbonate system is the pH (acid-base) buffer of seawater" (cms.udel.edu). As the excess of carbon dioxide was added to the water, more hydrogen ions were released, which cause the calcium carbonate to become bicarbonate, which then helps the ocean water to become more basic, (to be exact a pH of 8.1), the level at which it should normally be.
Now, the argument of whether or not this experiment is a valid model of ocean acidification solely depends on what the goal of the experiment is. If one is trying to catch a glimpse of the general effects of carbon dioxide on the pH of the ocean, than this experiment works quite well. On the contrary, if one is trying to see the actuality of how carbon dioxide affects the ocean and its pH, this experiment does not quite fit the bill. For one, a person simply blowing into a balloon and the CO2 escaping through a straw into the ocean water will add quite a different percentage/amount of carbon dioxide into the water than would happen in the environment. The sole fact that the beginning pH of both waters was much lower than that of real world waters simply shows just how high the percent error can be in this particular experiment. Also, there was most likely saliva and rubber residue in the air that came out of the balloon which could have also effected the ph.
All inclusive, ocean acidification is dramatically increasing because of human related increases in carbon dioxide emissions, which makes it an important topic in our AP Environmental Science class because the emitted CO2 is mainly originated by humans and is having major effects on our Earth's overall well being. For instance, a Smithsonian Ocean Portal article and a Climate Interpreter article examine and scrutinize the impairment of ocean acidification on aquatic ecosystems and species, "so much carbon dioxide is dissolving into the ocean so quickly that this natural buffering hasn’t been able to keep up, resulting in relatively rapidly dropping pH in surface waters" (Smithsonian Ocean Portal) and "a more acidic environment has a dramatic effect on some calcifying species, including oysters, clams, sea urchins, shallow water corals, deep sea corals, and calcareous plankton" (PMEL, NOAA). Each article continues on to explain the overall impact that ocean acidification has on overall marine life and each helps in embellishing this realization. In the grand scheme of things, this experimental lab assists in showing that carbon dioxide does indeed have a discrete effect on the pH of water however, it fails to exhibit the attested span of the problem.
Citation(s):
"Lab 7: Ocean Acidification: Too Much of a Good Thing?" Climate and the Carbon Cycle. N.p., n.d. Web. 17 Apr. 2015. <http://serc.carleton.edu/eslabs/carbon/lab7.html>.
"Ocean Acidification." Smithsonian Ocean Portal. N.p., n.d. Web. 17 Apr. 2015. <http://ocean.si.edu/ocean-acidification>.
"Ocean Acidification." Ocean Acidification. N.p., n.d. Web. 17 Apr. 2015. <http://www.pmel.noaa.gov/co2/story/Ocean%2BAcidification>.
"Ocean Acidification." Ocean Acidification. N.p., n.d. Web. 17 Apr. 2015. <http://co2.cms.udel.edu/Ocean_Acidification.htm>.
By: Noah Nelson